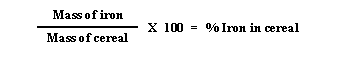Mixtures and solutions
http://walledlake.k12.mi.us/maple/MixturesSDefinitions.htm
http://galileo.phys.virginia.edu/education/outreach/8thgradesol/MixSoln.htm
Definitions for Mixtures and Solutions
matter:
all
materials in the universe.
mixture:
two or more
materials mixed or stirred together.
dissolve:
the process of a
material mixing with a liquid in a special way.
When salt mixes
with water, it seems to disappear and
cannot be seen
until the water dries up.
solution:
a special mixture formed when a material
dissolves in a liquid
and cannot be filtered out.
saturation:
a solution is saturated when as much
solid material (such as
sugar) as possible has dissolved in
a liquid (such as coffee),
and any additional solid material
sits on the bottom.
property:
a characteristic of an object; something
you can observe such as
size, color,
shape, or texture.
evaporation:
to dry up; to change from a liquid
into a gas.
soluble:
the property that substances have
of dissolving in solvents, such as
the soluability
of salt in water.
saturated
solution:
a solution in which no more
material will dissolve; additional
material settles to the bottom.
concentration:
the amount of material dissolved in
a measure of liquid; the more
material dissolved in the liquid, th more concentrated the solution.
dilute:
to make a solution less
concentrated, usually by adding more
liquid.
volume:
the three-dimensional space
occupied by something.
chemical
reaction:
a change that occurs as a result of
mixing two or more materials
together. A reaction results in the
formation of new materials
with different properties from the
original ones.
All
materials should be available for each group of students (workstation). The
sand, salt, iron filings and cooking oil can be in cups or small beakers. Go
over the background information with the students. Show them examples of
mixtures, solutions, and suspensions, and go over any new vocabulary words.
Explain
that in the following activity, each group has to make two mixtures using three
ingredients each from the list of ingredients in the procedure. One of the
mixtures must contain water; the other does not. After making the mixtures,
each group must come up with a procedure for separating the ingredients.
In
part #2, the students have to grind Total Cereal into a powder and determine
the percentage of iron in the mixture. Have a cup of cereal, magnet, scrap
paper, empty plastic cup and balance available at each station.
Properties of Mixtures and
Solutions
Objectives
Students
will
- identify and separate the
components in mixtures;
- determine the iron content in
cereal;
- determine the concentrations of
components of a mixture;
- use the following skills: making
and applying observations, organizing and analyzing information, and
measuring and recording data.
Starting the activity
Materials
- 4 g salt
- Two beakers (at least 500 mL)
- 4 g calcium carbonate or flour
or talcum powder
- stirring rod
- flashlight
- water
Procedure
1. To illustrate the difference between
a solution and a suspension, add about 4 grams of salt to 500 mL of water. Also, prepare a solution of calcium carbonate
(4.0 grams) and 500 mL of water.
2. Make certain that each mixture is
stirred well. You have to list any differences or similarities between the two
mixtures.
3. Darken the room and shine a
flashlight through each beaker. The beaker with calcium carbonate will appear
cloudy. Point out that this method is one way to differentiate between a
solution and a suspension.
The
particles in a suspension are larger and more unevenly distributed than those
of a solution. When light passes through the suspension, the larger suspended
particles scatter the light in all directions producing a cloudy appearance.
This phenomenon is known as the Tyndall
Effect. Most students have experienced the same effect in a movie theater, when light from the projector becomes visible
through the dust in the air.
Background Information
Mixtures are combinations of
substances that are not combined chemically. The amounts of substances in a mixture can
vary. Students should understand that mixtures can change in appearance but
this does not mean that any substances have changed in their chemical
composition. Mixtures can be
combinations of elements or compounds. Most substances found in nature
are mixtures. A pure element or a pure compound is rarely found. Mixtures can
be in any of the four phases of matter- or they can be in combinations of
different phases. Air is a mixture of gases, milk is a mixture of solids and
liquids, alloys are mixtures of solids. Mixtures
that do not appear to be distributed the same throughout are said to be heterogeneous,
and those that are the same throughout are called homogeneous.
Solutions are common types of
homogeneous mixtures. Sugar and water form a solution when mixed. The sugar becomes evenly
distributed throughout the solution, so that one portion is not sweeter than
another. The dissolved portion of the
solution is called the solute (sugar) and the dissolving portion is the solvent
(water). If more sugar is added to the solution, the entire solution becomes
sweeter and we say that it is more concentrated. In solution, sugar and water
have not lost their properties, only combined them. Pouring a solution, like
sugar and water, through filter paper will not separate the mixture,
the sugar particles are too small. The best method is distillation; the water
evaporates and the sugar is left behind.
All
materials are not soluble. A suspension is a mixture in which the solute
particles are larger than molecules or ions. Particles this size cannot adhere
to the molecules of the solvent and will eventually settle out. The particles
in a suspension are in the range of 10-2000 angstroms in diameter (1A =
.00000001 cm). The particles in a solution are usually much less than 10 A. As
a result, in addition to settling, the particles in a suspension scatter light
when it is passed through, giving it a cloudy appearance (Tyndall Effect).
![]()
Student Activity
Materials
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Procedure - Part 1: Making and Separating Solutions
1.Use three ingredients from the following
list to include in your first mixture: sand, salt, water, iron filings and
cooking oil. Enter the name and physical description of the ingredients that
you chose in the data sheet.
2.Mass out 2.0 grams of each solid and
15 mL of water if needed.
3.Mix the ingredients in the beaker
and stir with the stirring rod. Record the description of the mixture in the
table.
4.Write out a procedure for separating
the mixture in the data sheet. Follow your procedure and separate the mixture.
Save all the parts of the mixture and show them to your teacher.
5.Repeat the procedure in steps 1-4
for your second mixture.
Data Sheet
|
Mixture
1
Description of Mixture Procedure for Separating Mixture #1 1._____________________________________________________________________ 2.____________________________________________________________________ 3.____________________________________________________________________ 4.____________________________________________________________________ Mixture 2
Description of Mixture Procedure for Separating Mixture
#2 1._____________________________________________________________________ 2.____________________________________________________________________ 3.____________________________________________________________________ 4.____________________________________________________________________ |
Procedure - Part 2: Determining the Percentage of Parts of a Mixture
- Place an empty cup on the
electronic balance, press "tare" to remove the mass of the cup.
- Fill a 100 mL
beaker with the breakfast cereal. Transfer the cereal, a little at a time,
to a mortar and grind it to a powder. Transfer the ground cereal to the
cup. When you are finished grinding all the cereal from the beaker and
transferring it to the cup, find the mass. Enter the results in the table.
- Place the magnet into the
ground cereal and stir it around several times to collect the reduced
iron. Remove the magnet and rub off the particles that stuck to it onto a
piece of scrap paper. Repeat this procedure several times to insure you
have collected as much iron as you can. Remove any items that are clearly
not magnetic.
- Mass the cup and contents again
and record the mass in the table.
- Subtract the mass of the cup
and contents after the iron was removed from the original mass of the cup
and contents to find the mass of the iron that was removed. Enter the
value in the table.
- Find the percentage of iron in
the cereal by dividing the mass of the iron (#5) by the original mass of
the cereal (#2) and then multiply by 100.
|
|
Data Table
|
Original Mass of Cereal (g) |
|
|
Mass of Cereal After Iron is Removed (g) |
|
|
Mass of Iron (g) |
|
|
Percentage of Iron Contained in Cereal |
|
Extensions
1.
A good extension or even an assessment would be to give each group an unknown
mixture or solution and have them determine the ingredients. You could use either
the substances given here or others.
2.
Students can investigate how salt depresses the freezing point of water. They
can design their own experiment or follow the procedure below.
- Fill a 250 mL
beaker 2/3 full of crushed ice. Add water to fill up the spaces between
the ice. Stir well. Measure and record the
temperature.
- Add 5 grams of salt (NaCl) to the ice water mixture and stir well to
dissolve the salt. Measure and record the temperature.
- Predict what will happen when a
second 5 grams of salt is added to the solution. Add the salt (total 10
grams). Stir to dissolve and record the temperature.
- Add another 5 grams (total 15).
Stir and dissolve. Measure and record the temperature again.
- Keep adding salt, 5 grams at a time,
dissolving and recording the temperature each time.
- Make a data chart and graph
Grams of Salt vs. Temperature.
![]()
Assessment
1. How do the properties of a mixture
differ from the properties of the components of the mixture?
2. In terms of physical properties,
what is there about mixtures that makes it possible to
separate out the components?
3. Describe in detail the steps you
would take to find the percentage of salt in a sample of salt water.
Answers to Assessment
1. The chemical properties of the
components of a mixture do not change within a mixture. The properties of the
whole mixture vary greatly depending on the amount of each component and the
specific property being investigated.
2. The components of a mixture are not
chemically combined; therefore, the individual chemical and physical properties
of the two components remain individual and unique, allowing for easy
separation based on these properties. Separating a mixture can be likened to
separating two different colored marbles from a jar.
3. Step one: Find the initial volume and mass of
the sample. Use a graduated cylinder and a balance (be sure to scratch the mass
of the cylinder).
Step two: Heat the solution gently in a flask with a stopper that has
tubing running through it and into a separate flask. Continue until all water
is gone and only salt remains.
Step three: Put the remaining wet salt onto a Buchner funnel with pre-massed filter paper over top the
second collection flask. Vacuum filter the sample of salt.
Step four: Weigh the filter paper and subtract
the actual mass of the paper for the mass of the salt. Find the volume of the
separated water. Use the density to find the mass (the two values should be
equal).
Step five: Calculate the percentage of salt to
water by dividing the mass of salt by the mass of water and multiplying by 100.