CHEMICAL REACTIONS
GROUPS OF METALS
Extracted from Chemistry for You (Lawrie
Ryan. Published by Nelson Thornes,
2nd ed. 2001)
►Group
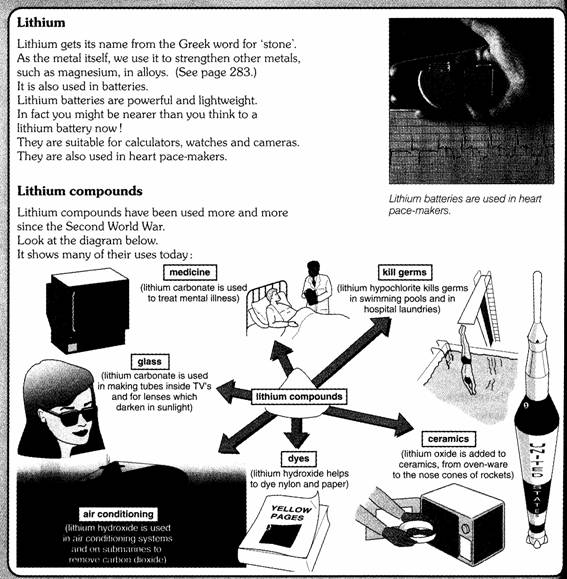
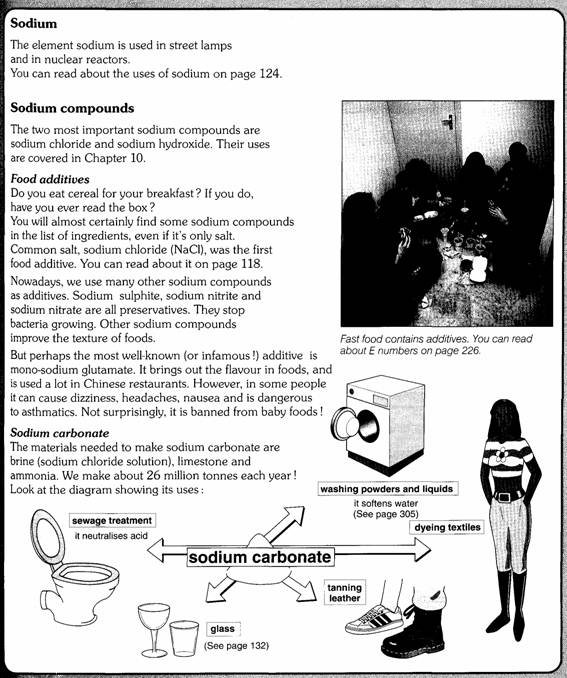
1 The alkali
metals
The elements in this first group don’t have many uses
as the metals themselves. They are too reactive. However, you will certainly
use some of their compounds every day.

 Demonstration
5.1 Looking at sodium
Demonstration
5.1 Looking at sodium
Your teacher will show you a piece of sodium.
What is it stored under? Why?
Does the sodium look like a metal?

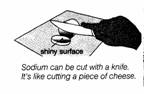 Your
teacher will cut a piece of sodium with a knife.
Your
teacher will cut a piece of sodium with a knife.
• How soft is it? What does it look like inside?
• Is this more like a metal now?
Your teacher will warm a small piece of sodium gently
on a combustion spoon.
• How easily does sodium melt?
Now you can see why sodium, and the other alkali
metals, are unusual metals. They have low melting
points and are very soft. For metals, they also have very low densities. You
will see in the next experiment that lithium, sodium and potassium float on
water!
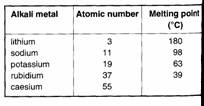

Look at the table opposite:
• Can you see a pattern going down the group?
• Can you predict the melting point of caesium?
Typical metals have much higher melting points. For
example, iron melts at 1540 °C.
 Reactions
of the alkali metals
Reactions
of the alkali metals
The alkali metals are the most reactive group of
metals in the Periodic Table. They are too dangerous for you to use in
experiments. However, your teacher can show you some reactions of lithium,
sodium and potassium.

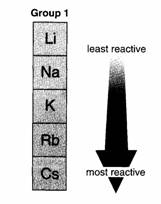
The Periodic Table is very useful. Its groups make
chemistry easier! You only have to learn the reactions of one element in a
group. The others are usually similar. For example,
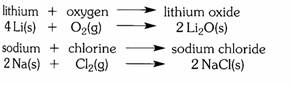
lithium +wateràlithium hydroxide+ hydrogen
(alkaline solution)
![]()
Knowing this, we know the equations for the other
alkali metals:
sodium
+ water » sodium hydroxide + hydrogen
![]()
• Can you write a word and symbol equation for
potassium reacting with water?
![]()
Other reactions
The alkali metals react well with non-metals. For
example,