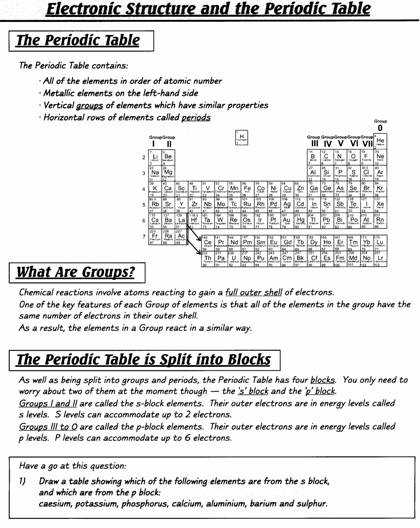
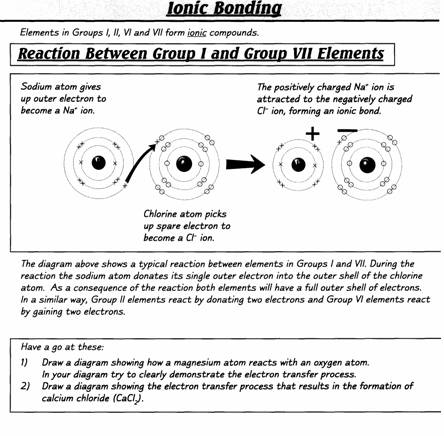
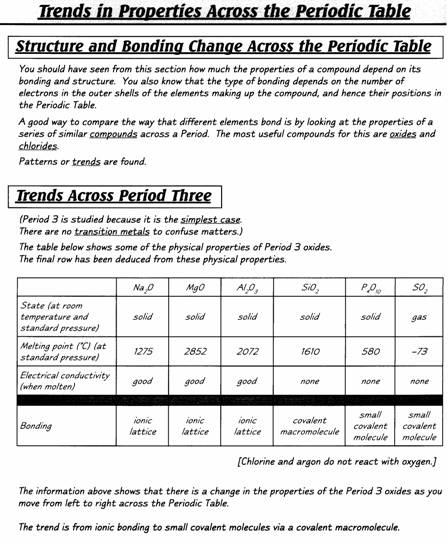
INTRODUCTION
Extracted
from Chemistry for You (Lawrie Ryan. Published by Nelson Thornes, 2nd ed. 2001)
History of the atom
The word atom comes
from a Greek word meaning something which can’t be split.
This fits in nicely
with
In the late 1800s and
early 1900s, scientists had to think up new pictures of atoms to explain new
observations.
For example, in 1897
J.J. Thomson put forward his ‘plum
pudding’ theory. He thought atoms were balls of positive charge with tiny
negative particles stuck inside. The negative particles were called electrons.
He said they were like the currants in a bun or Christmas pudding.
This model explained
Thomson’s experiments with electricity very well. However, later experiments
using radioactive particles needed a new picture. By 1915, scientists, like
Ernest Rutherford and Niels Bohr, had
developed a model that is still useful today.
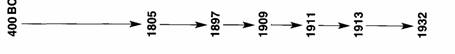
Democritus suggests that all things are made of
particles.
John Dalton’s atomic theory. Atoms of the same element are all alike. They combine
to make compounds.
J.J. Thomson finds the electron.
Ernest Rutherford discovers the proton.
Ernest Rutherford discovers the nucleus.
Niels Bohr suggests that electrons are found in shells
around the nucleus.
James Chadwick proves that neutrons exist.
Inside the atom
There are 3 types of
particle inside an atom. These are protons,
neutrons and electrons.
The protons and
neutrons are found squashed together in the middle of the atom.
The middle, called the
nucleus, is incredibly small and dense.
The tiny electrons whizz around this nucleus.
Protons are positively
charged. Electrons are negatively charged.
Neutrons have no
charge. They are neutral.
Protons and neutrons
are the heavy particles in an atom.
They each have a mass
of 1 atomic mass unit, and are found in the nucleus (centre) of an atom.
We can ignore the tiny
mass of the electrons.
The electrons orbit
the nucleus in shells.
The 1st shell can hold 2 electrons
The 2nd shell can hold
8 electrons, as can the 3rd shell.
The atomic number (Z)
= the number of protons (which equals the number of electrons).
The mass number (A) =
the number of protons + the number of neutrons.
Isotopes are atoms
with the same number of protons, but different numbers of neutrons.
ACTIVITIES
1. Copy and
complete: chemical, physical, atom, molecule, compound, atoms, simpler
a) The smallest part
of an element is called an …………………All the.................... in an element are the same. Elements can’t be broken down
into ................... substances.
b) Atoms joined or
bonded, together chemically are called……………. If a substance is made from more
than one type of atom, it is called a ………………
c) In
a.................... change, new substances are
formed. However, no new substances are made in a ………………..change.
2. Look at the boxes
below:

Which box contains:
a) one
element
b) a
mixture of elements
c) a
pure compound
d) a
mixture of elements and a compound? What might be happening in this box?
3. Complete the year with
the name of the scientists and their contributions:
4. Copy and complete:
There are 3 types of
particle found inside atoms:a)…………..
b)……………...and c)……………..
This table shows their
mass and charge:

The protons and
neutrons are found in the…………….. of the atom, called
the nucleus.
The
................. zoom around the nucleus in
shells. The 1st shell, which is .................. the nucleus, can hold....
electrons, whereas the 2nd and 3rd shells can hold ...... electrons.
5. a) What is the atomic number of an atom?
b) What is the mass number of an atom?
c) What is the atomic
number of the atom below? What is its mass number?
a) What is the atomic
number of an atom?
b) What is the mass
number of an atom?
c) What is the atomic
number of the atom below? What is its mass number?
![]()
6. Give the numbers of
protons, electrons and neutrons in the atoms below:
a) 147N; b) 2010Ne;
c) 199F; d)3919K;
e) 6027Co; f)23592U
7. Draw fully labelled
diagrams of the atoms below:
a) 42He;
b) 94Be; c) 2713Al; d)4020Ca;
8. Copy this table and
fill in the gaps.
Use Periodic Table to
help you

Do you have to look up
the atomic number of lithium, given the information in the table above? Explain
your answer.
9. Hydrogen (atomic
number 1) has 3 isotopes.
They can be shown as 11H,
21H and 31H.
a) What are isotopes?
b) What is the
difference between the 3 isotopes?
c) Hydrogen reacts
with chlorine in sunlight, forming hydrogen chloride:
H2+ Cl2
→ 2HCI
Would you expect the
same reaction for each isotope of hydrogen? Why?
d) Chlorine exists
naturally as 2 isotopes. 75% is 3517Cl, and 25% is 3717Cl.
Show why the relative atomic mass of chlorine is 35.5.
e) The element
chlorine is a gas. Its formula is Cl2 How many different masses of
the Cl2 molecule would you expect to find in a sample of chlorine
gas? Explain your answer.
10.Work
out the relative formula mass of:
a)H2O b)
C2H5OH c)
Na2SO4
(R.A.M.s: H = 1, O= 16, C = 12, Na = 23, S=32)
IN THE LABORATORY
www.chemistry.lmt.md/sezennur/
subjects/experiments/018_flame_test.pdf
Flame
Test
PRE-LAB DISCUSSION
The normal electron configuration
of atoms or ions of an element is known as the “ground state.” In this most
stable energy state, all electrons are in the lowest energy levels available.
When atoms or ions in the ground state are heated to high temperatures, some
electrons may absorb enough energy to allow them to “jump” to higher energy
levels. The element is then said to be in the “excited state.” T
This excited
configuration is unstable, and the electrons “fall back to their normal
positions of lower energy. As the electrons return to their normal levels, the
energy that was absorbed is emitted in the form of electromagnetic energy. Some
of this energy may be in the form of visible light. The colour of this light
can be used as a means of identifying the elements involved. Such crude
analyses are known as flame tests.
Only metals, with
their loosely held electrons, are excited in the flame of a laboratory burner.
Thus, flame tests are useful in the identification of metallic ions. Many
metallic ions exhibit characteristic colours when vaporized in the burner
flame. In this experiment, characteristic colours of several different metallic
ions will be observed, and an unidentified ion will be identified by means of
its flame test.
PURPOSE
Observe the
characteristic colours produced by certain metallic ions when vaporized in a
flame. Identify an unknown metallic ion by means of its flame test.
EQUIPMENT
graduated cylinder, 10-mL wire
loop
platinum laboratory
burner
glass-marking pencil test
tubes
test tube rack
MATERIALS
HCI(conc.)
Unidentified solutions
0.5 M solutions of nitrates of: Na+,K+, Ca+2, Sr+2, Ba+2,
Cu+2
PROCEDURE
1. Measure 5 mL of tap water in a graduated cylinder and pour the water
into a 13 x 100 test tube. Using a marking pencil, mark the outside
of the tube to indicate the level of the water. Discard the water. Using
the marked tube as a guide, mark seven clean test tubes at approximately the
same level. Place the clean tubes in a test tube rack. Set the other test tube
aside.
2. Into each of the
clean test tubes, pour 5 mL of a different nitrate
solution. Mark each test tube to indicate the metallic
ion it contains.
3. Pour about 10 mL of concentrated hydrochloric acid into a 50-mL beaker. CAUTION:
Use extreme care in handling this acid. To clean the wire loop, dip the loop in
the acid and then heat the loop in the outer edge of the burner flame. Continue
to clean the loop in this manner until no colour is observed in the flame.
4. Dip the clean wire
loop into one of the nitrate solutions. Place the loop in the outer edge of the
burner flame and move the loop up and down (Figure). Note the colour in the
flame. Record your observations in the data list provided.
 5. Clean the wire loop
as described in step 3. Repeat step 4 using a different nitrate solution.
5. Clean the wire loop
as described in step 3. Repeat step 4 using a different nitrate solution.
6. Test each nitrate
solution in the same manner, cleaning the loop thoroughly between tests. Record
all your observations in the data list.
7. Obtain a sample of
an unknown solution. Perform a flame test and identify the metallic ion present
by the colour of the flame.
OBSERVATIONS AND DATA
Metallic Ion Color in Flame
Na+ ____________________
Ca+2 ____________________
Sr+2 ____________________
Ba+2 ____________________
Cu+2 ____________________
Unknown ____________________
CONCLUSION AND QUESTIONS
1. What inaccuracies
may be involved in using flame tests for identification purpose?
2.Which pair of ions produce
similar colours in the flame tests?
3.Explain how the colours
observed in the flame tests are produced.
4.Define these terms:
a. quanta
b. ground state
c. excited state
5. What is a
spectroscope? What is observed if the flame tests are viewed through a
spectroscope?
http://www.qonulevi.com/chemistry