THE PERIODIC TABLE
Extracted from Chemistry for You (Lawrie
Ryan. Published by Nelson Thornes,
2nd ed. 2001)
Sorting out
the elements
Scientists
like to find patterns.
Around 200
years ago, scientists were discovering lots of new elements. However, they
struggled to find any links between the different elements.
At the time,
some substances, which were thought to be elements, were in fact compounds.
Other
elements had not yet been discovered.
No wonder
finding a pattern was tricky!
Finding the
pattern
Real
progress was made around 1865 by John Newlands.
He put the
elements in order of their atomic mass.
He found that
every eighth elements was similar.
 Unfortunately,
his pattern only worked for the first 15 elements known at that time. After
that, he could see no links between the rest of the
elements. Other scientists made fun of his ideas. They suggested that he could
have done better by sorting the elements into alphabetical order!
Unfortunately,
his pattern only worked for the first 15 elements known at that time. After
that, he could see no links between the rest of the
elements. Other scientists made fun of his ideas. They suggested that he could
have done better by sorting the elements into alphabetical order!
In 1869 the
problem was solved by a Russian called Dmitri Mendeleev.
He also
tried putting the elements in order of their atomic mass.
He made a
table of elements.
New rows
were started so that elements which were alike could line up together in columns.
He wanted a
table of regular (periodic) patterns.
However, Dmitri was not afraid to take risks. When
the pattern began to go wrong. he would leave a
gap in his table. He claimed that these gaps were for elements that had not yet
been discovered. He even changed the order round when similar elements didn’t
line up.
As you might
expect, people doubted his “Periodic Table”.
However. he used his table to predict the properties of elements which
could fill the gaps.
In 1886, the
element germanium was discovered. The new elements matched Dmitri’s
predictions.
Finally
other scientists accepted his ideas.
►The
Periodic Table
Although Mendeleev’s table was accepted, there was one thing that he
could not explain.
Why did he
sometimes need to change the order of atomic masses to make the pattern carry
on? The answer lies inside the atoms. The atoms of elements in the Periodic
Table are not arranged in order of mass. It is their
number of protons (atomic number) which really matters.

Groups
 There
are 8 groups in the Periodic Table.
There
are 8 groups in the Periodic Table.
A group is a
vertical column.
All the
elements in a group have similar properties.
They are a
chemical family
Look at the
Periodic Table above:
Some groups have
‘special family’ names.
Can you find
Group 7?. What is the group called?
Nave you met
any elements from this ‘family’ before’?
Other groups
are just known by their group number.
Groups are
families of elements. The members of the family are similar but not exactly the
same
Notice that
the transition metals form a block on their own.
Periods
Periods
are the rows across the Periodic Table.
You read the
table like a book. Start at the top. and work your way
down, reading from left to right. So there are 2 elements in the 1st period. H and He.
The 2nd
period has 8 elements. starting with Li.
 Which
is the last element in the 2nd period?
Which
is the last element in the 2nd period?
Can you
count how many elements are in the 3rd period
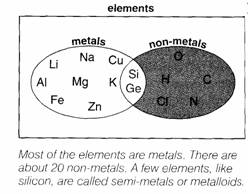
►Metal
and non-metal elements
92 elements
are found naturally on Earth.
Most can be
sorted into 2 sets — the metals and the non-metals.
Let’s look
at their properties.
Metals
Over three
quarters of the elements are metals. The experiments below show us some typical
properties of metals.

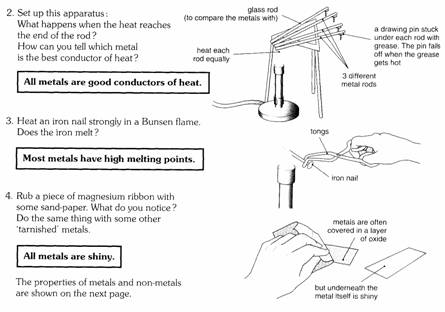
Properties
of metals
A!! the properties shown below are physical properties. They
describe the metal itself, not its chemical reactions.
Metals are
important in all our lives. Can you think of a use to match each property shown
below?

Some exceptions
Not all
metals have these properties.
For example,
the alkali metals (lithium, sodium, potassium) are soft metals. They have low
densities and have low melting points.
Mercury also
has a low melting point for a metal.
 ►Metallic
bonding
►Metallic
bonding
Think of
some of the things around your home that are made of metal.
Did you
include all the wiring, any radiators, your hot-water tank, or your cutlery and
pans? Do you know which metals these things are made from? Which properties
make metals good for these uses?
We have
looked at ionic and covalent bonding. But the atoms in a metal are held
together in a different way.
Do you
remember all the properties of metals?
Any ideas we
have about the bonding and structure of metals must be able to explain their
properties.
In general,
metals:
•
have high melting and boiling points
•
conduct electricity and heat
•
are hard and dense
•
can be hammered into shapes (they are malleable)
•
can be drawn out into wires (they are ductile).
We believe
that metal atoms (or ions) are held together by a ‘sea’ of electrons. Look at
the diagram below:
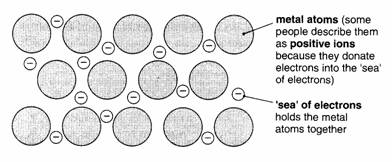
Each metal
atom gives up electrons from its outer shell into the ‘sea’ or ‘cloud’ of
electrons.
The
electrons can drift about in the metal. These free electrons explain how
electricity can pass through solid metals.
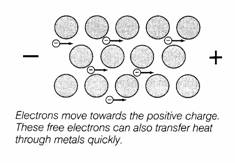 What
happens when one end of the metal is made positive and
the other end negative?
What
happens when one end of the metal is made positive and
the other end negative?
Properties
of non-metals
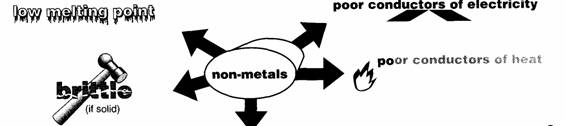
Some exceptions
Carbon in
the form of graphite is a good conductor of electricity. Carbon in the form of
diamond has a very high melting point. Can you find out about any other
‘exceptional’ elements?
►Oxides
of metals and non-metals
You have
seen the physical properties of metals and non-metals. Now let’s look at some
chemical properties. The chemical properties of a substance describe its
reactions.
We can make
oxides by reacting elements with oxygen.
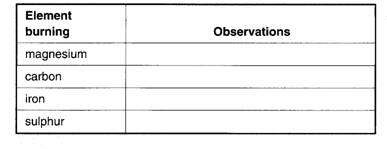
 Experiment
4.2 Burning elements in oxygen
Experiment
4.2 Burning elements in oxygen
Collect 4
large test-tubes of air.
Use tongs or
a combustion spoon to heat your element in a Bunsen flame. Then plunge it into one
of your tubes of oxygen gas.
When the
reaction has finished, add a few drops of universal indicator solution. Then
shake the tube gently.
Record your
results in a table:
• Which
oxide is slightly alkaline?
• Which
oxides are acidic?
The
elements burn more fiercely in pure oxygen than in air.
This is because only about 20% of the air is made up of oxygen.
Metals
burning
The metal
oxides made are solids. For example,
magnesium
+ oxygen à
magnesium oxide
![]()
Can you
write the word equation for iron reacting with oxygen?
Non-metals
burning
When
non-metals burn, their oxides are often gases. For
example,
carbon
+ oxygen à
carbon dioxide
![]()
Try to write
the word and symbol equations for sulphur reacting with oxygen.
(The product
is sulphur dioxide gas. Its formula is SO2)
Basic and
acidic oxides
In the next
experiment, you can test the pH of more oxides.
The oxides
have been dissolved in water for you.
Experiment
4.3 Testing the pH of oxides
Collect 2 cm
of each oxide solution in separate test-tubes.
Add 3 drops
of universal indicator solution to each oxide solution.
Record your
results in a table like the one below:

Which
compounds in the table are metal oxides?
What do you
notice about your results for the metal oxides?
What can you
say about your results for the non-metal oxides?
 Metal
oxides
Metal
oxides
If a metal
oxide dissolves in water, it makes
an
alkaline solution. For example,
sodium
oxide + water àsodium
hydroxide
![]()
Some metal
oxides do not dissolve in water. Iron oxide is insoluble. These metal oxides
test neutral with universal indicator.
However,
they do react with acid. Insoluble metal oxides are called bases. Look at the
diagram opposite:
Non-metal
oxides
Most
non-metal oxides dissolve in water to form acids. For example,
carbon
dioxide + water àcarbonic
acid
![]()
One
exception is the oxide of hydrogen.
What do we
call this oxide ? What is its pH?
However, in
general we can say:

Summary
•
The Periodic Table arranges the elements in order of atomic number.
•
Elements with similar properties line up in vertical columns. These columns are
called groups.
•
There are 8 groups in the Periodic Table.
•
A row across the Periodic Table is called a period.
•
The elements can be divided into metals and non-metals (with a few semi-metals
or metalloids in between).
•
Metals are good conductors of heat and electricity. They are shiny, malleable
(can be hammered into shapes) and ductile (can be drawn out into wires). Most
are hard, dense and have high melting points.
•
Iron, cobalt and nickel are the magnetic metals.
•
Most non-metals are gases. They have low melting and boiling points. They are
poor conductors of heat and electricity. If solid, they are usually dull and
brittle.
•
In general, metal oxides are basic.
Non-metal oxides are usually acidic.
►Questions
1. Copy and
complete:
The elements
line up in order of atomic.......... in the Periodic Table. There are .... groups. The elements in each
group have.... properties.
Groups form
v_ _ _ _ _ _ _ columns, whereas periods are h_ _ _ _ _ _ _ _ _ rows. Most of
the
elements
can be divided into 2 sets - the…………….and the non-metals. In general, metal
oxides are ………...., and non-metal oxides are…………………
2. Copy and
complete this table:

3. Not ah
metals and non-metals have the properties listed in question 2. Can you name
some exceptions and say why they are unusual?
4. Yasha and Eric tested solutions of some unknown oxides.
Look at their results:

D did not
dissolve in water, but did dissolve in dilute acid.
a) Which are
the oxides of non-metals? How can you tell?
b) Which
metal oxide is an alkali and which is a base? Explain your answer.
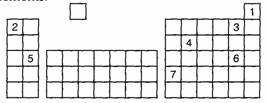 5.
The numbers in this Periodic Table represent elements.
5.
The numbers in this Periodic Table represent elements.
a) Which 2
elements are in the same group? Give the name and number of this group.
b) Which
elements are in the 2nd period?
c) Which
elements are metals?
d) Which
group is element 7 in? Which period is it in?
e) Which
element is a semi-metal (or metalloid)?